NEWS
HIV-1 Solution v2 - NGS analysis for drug resistance detection on Reverse Transcriptase, protease, integrase and gp120 regions.
Antiretroviral therapy is subject to continuous evolutions and improvements because today people who are treated for HIV live longer and with a better quality of life. In the individual, HIV virus can reproduce very quickly by introducing mutations during the reproduction process, mutations which increase the virus ability to replicate even during the administration of antiretroviral drugs against which the virus acquires resistance. Due to the increasing incidence of resistant viruses in the population, drug resistance is one of the main concerns of those who must treat HIV.
The availability of a standardized test for genotyping and evaluating drug resistance in the clinical setting has a key role for the success of anti-HIV therapy, whose primary goal is essentially to increase the quality of life of patients by decreasing the viral load for as long as possible, protecting or promoting the recovery of the immune system and reducing the occurrence of diseases related to immunodepression.
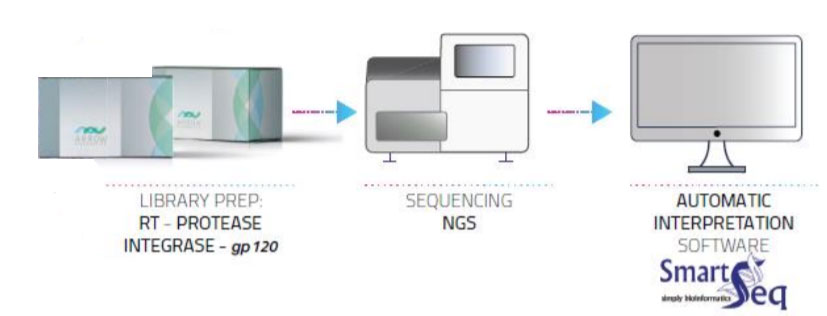
 HIV-1 Solution v2 is an in vitro molecular test in Next Generation Sequencing (NGS) based on the well-known and scientifically proven amplification by RT-PCR (“Reverse transcriptase-polymerase chain reaction”) of the viral RNA regions considered as having a key role for drug development resistance (RT, Protease, Integrase and gp120).
HIV-1 Solution v2 is an in vitro molecular test in Next Generation Sequencing (NGS) based on the well-known and scientifically proven amplification by RT-PCR (“Reverse transcriptase-polymerase chain reaction”) of the viral RNA regions considered as having a key role for drug development resistance (RT, Protease, Integrase and gp120).The amplification of RNA by RT-PCR, starting from a biological sample (after extraction of RNA by standard procedures) and the subsequent sequencing in NGS, allow to identify the HIV genomic mutations (in regions which confer resistance against certain types of anti-retroviral drugs).
After sequencing, the samples are analyzed automatically using a dedicated software which, by querying the common databases, returns a report with all the mutations identified and any drug resistance associated to the HIV virus.